After a couple of pages of preliminaries, we are finally ready to discuss some materials science! Whereas page 2 employed convex hulls to maximize profits in a hypothetical bike shop, page 3 applies the same formalism to determine thermodynamic stability and demonstrate how chemical compounds can decompose to minimize energy.
And the adventure is far from over! In fact, next time we’ll add a whole new dimension to our analysis! For now, be sure to check out “Further Resources” below the comic for more information.
To be continued…
Further resources:
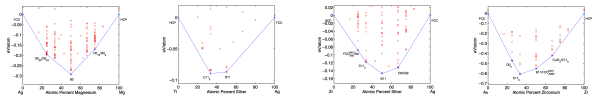
“Accuracy of ab initio methods in predicting the crystal structures of metals: A review of 80 binary alloys” by Curtarolo et al.
This (somewhat epic!) paper contains data for 80 binary convex hulls computed with density functional theory. The results are compared with known experimental data and it is determined that the degree of agreement between computational and experimental methods is between 90-97%.
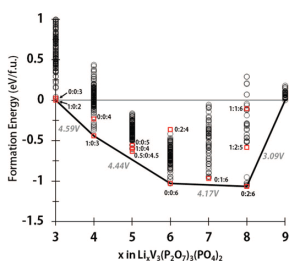
“A Computational Investigation of Li9M3(P2O7)3(PO4)2 (M = V, Mo) as Cathodes for Li Ion Batteries” by Jain et al.
The endpoints of a binary convex hull need not be elements. For example, in the Li ion battery field one often searches for stable intermediate phases that form at certain compositions as lithium is inserted into a framework structure. The paper above is just one example of many computational Li ion battery papers that use such “pseudo-binary” convex hulls.
“Configurational Electronic Entropy and the Phase Diagram of Mixed-Valence Oxides: The Case of LixFePO4” by Zhou et al.
Incorporating temperature into first-principles convex hulls is often possible, but not always straightforward or easy to do. Here is one example of this process that focuses on electronic entropy.
Materials Project Phase Diagram App
The Materials Project phase diagram app allows one to construct interactive binary convex hulls for any system by employing computed data on tens of thousands of compounds. You can also create phase stability diagrams for ternaries and higher!